The great horned owl (Bubo virginianus) is one of the most widespread and adaptable raptors in the Americas, occupying an enormous range of habitats from boreal forests to deserts and city suburbs. Its deep, resonant hoots and prominent ear tufts make it one of the continent’s most recognizable nocturnal birds, while its broad diet, powerful hunting adaptations, and tolerance of human-altered landscapes underpin its success.

| Common name | Great horned owl |
| Scientific name | Bubo virginianus |
| Alternative names | Tiger owl, hoot owl |
| Order | Strigiformes |
| Family | Strigidae |
| Genus | Bubo |
| Discovery | Described by J. F. Gmelin in 1788; widely known to Indigenous peoples of the Americas long before scientific classification |
| Identification | Large, powerful owl with prominent ear tufts, broad rounded wings, and a deep hooting voice; plumage variable but typically mottled brown with a barred underside and white throat patch; yellow eyes and massive talons |
| Range | Widespread from northern Alaska and Canada through the continental United States, Mexico, Central America, and much of South America to Tierra del Fuego |
| Migration | Generally non-migratory; most individuals remain year-round; northern birds show limited irruptive movements tied to prey cycles |
| Habitat | Occupies an exceptionally broad range of habitats: forests, woodlots, open country, deserts, grasslands, wetlands, and urban/suburban landscapes; favors mosaics of woodland and open areas |
| Behavior | Primarily nocturnal; solitary and territorial; hunts mainly from perches using acute vision and hearing; powerful predator; highly adaptable and tolerant of human-modified environments |
| Breeding | Monogamous; uses nests built by other birds, tree cavities, cliffs, or human structures; lays 1-4 eggs; only the female incubates; young branch at 5-6 weeks, short flights at 45-49 days; remain dependent for weeks to months |
| Lifespan | Typically 8-13 years in the wild; maximum wild record 28 years 7 months; maximum captivity record 34 years 10 months. |
| Diet | Broad generalist predator taking mammals, birds, reptiles, amphibians, fish, and invertebrates; diet dominated by mammals with significant regional variation |
| Conservation | Least Concern (IUCN); estimated around 5.7 million mature individuals; stable overall but subject to regional declines; main threats include secondary poisoning, vehicle collisions, electrocution, and occasional shooting |
Discovery
The great horned owl has been known to Indigenous peoples and early European naturalists in the Americas long before its formal scientific description. Its large size, penetrating voice, and conspicuous ear tufts gave it a prominent place in Indigenous cultures across North and South America, where it appeared in traditional stories, artwork, and ecological knowledge as a powerful nocturnal hunter. Early colonial observers also noted the species frequently, recognizing it as a characteristic owl of woodlands, prairies, and deserts throughout the continent.
The first formal scientific description was provided in 1788 by the German naturalist Johann Friedrich Gmelin, who assigned the species to the genus Strix and published the name Strix virginiana in his expanded edition of Linnaeus’s Systema Naturae. Gmelin’s diagnosis drew heavily on the work of the English naturalist George Edwards, who illustrated and described the owl in 1747 after examining a live bird brought from Virginia and several preserved specimens housed in prominent collections of the time.
Subsequent revisions of owl taxonomy led to the reclassification of the species into the genus Bubo, introduced by André Duméril in 1805. The current scientific name, Bubo virginianus, thus combines the Latinized reference to “Virginia” – the locality of Gmelin’s material, with the genus widely applied to large, tufted “eagle-owls.” The meaning of the Latin epithet reflects this early association with eastern North America, although the species proved to be far more widespread than initially recognized.
Modern phylogeographic work confirms that Bubo virginianus represents one of at least two major colonizing radiations of Bubo into the New World, likely spreading from ancestral Eurasian populations across the Bering land bridge. Historically, some authors speculated that the great horned owl and the Eurasian eagle-owl (Bubo bubo) might be conspecific owing to similarities in size and ecology, but molecular evidence does not support this view. Instead, recent genetic analyses place the snowy owl (Bubo scandiacus) as the probable sister species, with the great horned owl and lesser horned owl (Bubo magellanicus) forming a New World lineage distinct from Eurasian eagle-owls.
Within Strigiformes, broader molecular studies have reshaped the historical understanding of owl relationships. Owls were once linked to nightjars, but comprehensive multigene analyses show that the Strigiformes are instead sister to Coliiformes (mousebirds), forming a clade associated with trogons, kingfishers, rollers, and woodpeckers. Within the family Strigidae, the great horned owl belongs to the “eagle-owl” assemblage, which includes the genus Bubo together with a clade formed by Strix (including the former Ciccaba species) and likely Pulsatrix.
Fossil evidence suggests a long and stable presence of Bubo owls across North America. Pleistocene remains, found from the Pacific Northwest south into California caves and eastward to the Midwest and Georgia, fall within the modern distribution and indicate that ancient forms were often larger than today’s birds. One such fossil, described as Bubo sinclairi from Upper Pleistocene deposits in California, represents a robust paleosubspecies or closely related species larger than the living great horned owl.
Taxonomically, Bubo virginianus is highly variable, showing pronounced geographic differences in size and plumage that broadly follow ecogeographic rules such as Gloger’s and Bergmann’s. Historically, more than 20 subspecies names were proposed, though many reflect individual or clinal variation. Modern treatments typically recognize 14-15 subspecies, arranged in several regional groups (northern, North Andean, and tropical). Differences among these forms involve overall darkness or pallor, the intensity and pattern of ventral barring, the degree of tawny or gray coloration, and body size. Only one major taxonomic revision has occurred recently: the former southern Andean taxon B. v. magellanicus is now treated as a distinct species – lesser horned owl, based on consistent vocal, morphological, and genomic divergence.
The subspecies of the great horned owl represent broad geographic gradients rather than sharply bounded units, and their identification is often complicated by individual variation, wide-ranging movements, and intergradation in contact zones. These forms will be treated in greater detail in the range and morphology sections, but in brief, the northern and montane populations tend to be larger and paler, humid-coastal and tropical populations darker and smaller, and Andean forms distinctively dusky with heavy ventral markings.
Identification
The great horned owl is a large and powerfully built owl, measuring 46 to 63 centimeters (18.1 to 24.8 inches) in length with a wingspan commonly between 1.0 and 1.45 meters (3.3 and 4.8 feet), and reaching up to 1.53 meters (5 feet) in the largest individuals. Adults typically weigh between 0.91 and 2.5 kilograms (2 and 5.5 pounds), making it the second-heaviest owl in North America after the snowy owl and the heaviest resident owl in Central and South America. Its long, broad wings, massive head, and prominent ear tufts give it a distinctive silhouette both at rest and in flight, where it glides with wings held nearly horizontal.
In plumage, the great horned owl shows a complex mixture of brown, buff, gray, and black-lined patterns that vary with geography but follow a consistent overall plan. The facial disc is moderately defined and ranges from grayish in northern birds to warm buff or reddish brown in many southern and coastal populations. Large yellow eyes, framed by black and pale markings, dominate the face, and a conspicuous white throat patch flashes during calling. The upperparts are mottled and darker, while the underparts show pale buff to tawny tones crossed by variable dark barring. The wings are broad and rounded with barred coverts and banded primaries, and the tail shows 6-7 dusky bands.

Sexual dimorphism is expressed mainly in size rather than coloration. Females are noticeably larger and heavier than males across all subspecies. Broad samples show average masses of approximately 1.7 kilograms (3.8 pounds) in females and 1.3 kilograms (2.9 pounds) in males, though some regional forms differ markedly. A large female B. v. virginianus may exceed 2.5 kilograms (5.5 pounds), whereas males of smaller subspecies such as B. v. pacificus may weigh as little as 680 grams (1.5 pounds). Plumage pattern and coloration are essentially identical between the sexes, with no consistent differences in brightness, saturation, or markings.
Juveniles progress through several distinct plumage stages. Hatchlings are covered in dense white natal down, with closed eyes and inconspicuous bare parts. By the end of the first week, they develop a thicker coat of grayish to buff mesoptile down, often retaining patches of white on the tips. Between 2 and 3 weeks, the first sheaths of flight feathers begin to emerge, and small patches of developing ear tuft feathers may become visible.

Young in juvenile plumage appear softly textured and uneven, with fluffy contour feathers and a darker buff wash on the breast, faintly barred. The facial disc and white throat patch become better defined by 10-12 weeks, though the ear tufts remain shorter than in adults. The formative plumage, acquired gradually during late summer and autumn, resembles adult plumage except for smaller ear tufts, narrower juvenile tail feathers, and more sharply defined dark bars on the juvenile remiges and rectrices. Full adult appearance is typically achieved after several molt cycles, often not until the third or fourth basic plumage.
Subspecies and Geographic Variations
Across their wide range, great horned owls show pronounced geographic variation in size and coloration. Northern and interior forms such as B. v. subarcticus and B. v. heterocnemis tend to be paler, larger, and more buff-washed, with lighter feet and reduced rufous tones. Humid-coastal birds (B. v. saturatus, B. v. pacificus) are darker, grayer, and more saturated, with heavier ventral barring. Desert and dry-region forms (B. v. pallescens) are notably pale with reduced barring and immaculate or lightly marked feet. Tropical and Andean subspecies (B. v. mesembrinus, B. v. nigrescens, B. v. nacurutu) are generally smaller, richer rufous or darker brown, and often show heavier, more blotched ventral markings.
Vocalization
The great horned owl has an extensive and varied vocal repertoire, dominated by deep, resonant hoots but extending to a wide array of barks, screams, growls, whines, and chittering notes. Vocalizations differ between individuals and even within the same bird, with males producing the lowest, richest, and most far-carrying calls. Females, despite being larger-bodied, give slightly higher-pitched hoots due to a smaller syrinx. Territorial hooting is the most familiar sound: a rhythmic, multi-note sequence usually delivered with the bill closed and the body leaning forward, often described as “who-hoo-hoo,” “whoo-hoo” or similar patterns. Males and females frequently duet, their calls overlapping in long, synchronized exchanges that help reinforce pair bonds and advertise occupied territories.
Vocal development begins remarkably early. Owlets utter soft whimpering notes inside the egg, progressing after hatching to rasping chirps and weak begging calls. As they grow, their calls increase dramatically in volume and complexity. Juvenile males begin mimicking adult hoots during their first winter, though early attempts trail off into gasps or gurgling squawks; complete territorial hoots typically do not appear until late winter or early spring. Females develop hooting more abruptly, producing full female-type calls in their first spring. Throughout this period, young also give distinctive hawk-like screams that function as begging calls and contact notes.
Adults rely on an extensive set of non-hooting calls in social and defensive contexts. Both sexes produce sharp barking notes, piercing screams, hollow laughs, and catlike mews when disturbed, and nest defense often involves loud hissing, bill clapping, and guttural growls. Contact between adults and newly flying young is maintained through thin whistling notes, while soft chittering or conversational calls may occur between members of a pair. Many of these sounds carry well at night and contribute to the species’ vocal prominence across its range.
Vocal activity is most intense during the breeding season, often beginning one to two months before egg-laying. Calling peaks shortly after sunset and again before sunrise, with occasional bouts around midnight. Males usually vocalize from prominent perches near the nest site, while females may call from within the nest itself. This patterned, seasonally heightened soundscape, alternating deep territorial hoots with varied screams and barks, makes the great horned owl one of the most acoustically conspicuous nocturnal birds in the Americas.
Range
The great horned owl has an exceptionally broad distribution, occurring from the northern edge of the North American taiga to the southern tip of South America. In the north it reaches the treeline around 68°N, inhabiting western and central Alaska, the Yukon, the Northwest Territories, northern Manitoba and Ontario, northern Quebec, Labrador, Prince Edward Island, and Newfoundland. It is widespread across southern Canada and the United States, although local densities are lower in high-elevation portions of the Rockies and Appalachians and in the driest parts of the Mojave and Sonoran deserts.
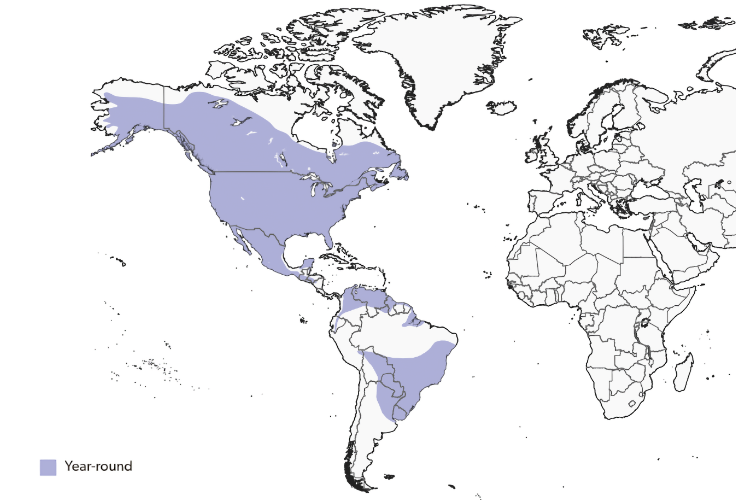
Through Middle America the species is resident across most of Mexico, extending south through the interior of northern Central America to western Nicaragua. It is absent from much of humid southeastern Mexico except the northern and central Yucatán Peninsula, and becomes very rare in Costa Rica, with only marginal occurrence in Panama.
In South America it is patchily distributed, absent from coastal mangroves of the Caribbean basin and scarce across Amazonia, but locally present through lowlands south to northeastern Argentina. In the Andes it ranges from Venezuela southward at elevations generally above 3,500 meters (11,500 feet), reaching over 4,300 meters (14,100 feet) in parts of Ecuador. Across this immense range, local occurrence is strongly tied to prey availability and suitable hunting structure rather than to vegetation type, making the species one of the most adaptable owls in the New World.
Subspecies and their ranges:
- B. v. algistus – western Alaska.
- B. v. lagophonus – central Alaska to northeastern Oregon and Montana.
- B. v. saturatus – coastal Alaska to northern California.
- B. v. pacificus – coastal California to northwestern Baja California.
- B. v. elachistus – southern Baja California.
- B. v. subarcticus – central Canada to the northern Great Plains.
- B. v. pallescens – interior California, Great Basin deserts, southwestern U.S., and western-central Mexico.
- B. v. pinorum – Great Basin and central Rocky Mountains.
- B. v. heterocnemis – Quebec, Labrador, and Newfoundland south to the Great Lakes.
- B. v. virginianus – southeastern Canada and eastern United States.
- B. v. mayensis – Yucatán Peninsula.
- B. v. mesembrinus – southern Mexico to western Panama.
- B. v. nigrescens – Andes of Colombia to northwestern Peru.
- B. v. nacurutu – eastern Colombia through the Guianas, northern and eastern Brazil, Bolivia, and northern Argentina.
- B. v. deserti – northeastern Brazil.
Migration
Great horned owls are non-migratory, and even the northernmost populations remain year-round on their territories. Individuals show strong site fidelity, and established adults generally remain in the same areas throughout their lives. Nevertheless, the species exhibits irruptive movements driven by prey cycles, particularly the periodic crashes of snowshoe hare populations in boreal regions. During such years, owls from Saskatchewan, Alberta, and the Yukon may undertake large southeastward movements, appearing in the Dakotas, Minnesota, Wisconsin, Nebraska, Kansas, and Iowa. These movements involve both juveniles and adults and represent opportunistic dispersal rather than true migration.
Long-distance dispersal can be substantial: the longest recorded movement exceeds 2,000 kilometers (1,250 miles), from Alberta to western Illinois. Most irruptive movements appear unidirectional, with little evidence that individuals return to their natal regions. Food availability strongly influences these patterns; non-breeding “floaters” are especially prone to disperse during low-prey years, while territorial adults tend to remain resident unless prey shortages are severe. In other parts of the range, smaller-scale seasonal shifts occur, including occasional northward movements from the Ohio Valley into Michigan, but these are irregular and limited in scope.
Habitat
The great horned owl occupies an exceptionally broad range of habitats, reflecting its adaptability and generalist hunting strategy. It occurs in deciduous, mixed, and coniferous forests across much of its range, but shows a marked preference for mosaic landscapes that combine woodland structure with open areas such as fields, wetlands, pastures, and agricultural land. These openings provide productive hunting grounds, while forested patches supply roosting sites and nesting substrates. The species readily uses secondary-growth woodlands, wooded swamps, orchards, riparian corridors, and forest edges, and it is one of the few large owls that regularly occurs in suburban parks and even urban environments when prey is abundant.
Across its northern and montane range, habitat use varies with local structure. In the boreal forest it is less common in extensive muskeg and dense conifer stands, instead favoring mixed and hardwood components. In the Rocky Mountains and interior West, it increases in areas of natural or anthropogenic clearing and frequently selects large stands of lodgepole pine or other conifers positioned near meadows or sagebrush openings. In deserts, occupancy depends on the availability of cliffs, rock outcrops, or scattered junipers for nesting and roosting. In the eastern United States the species uses oak- and elm-dominated forests, agricultural matrices, and fragmented rural landscapes where forest cover is interspersed with cropland and pasture.
Farther south, the species continues to exploit structurally diverse environments. In the Andes it is widespread at high elevations, typically from 2,500 to 4,500 meters (8,200 to 14,800 feet), using open montane valleys, shrublands, and barranca zones where hunting perches are available. In tropical regions it avoids dense mangroves and much of humid Amazonia but occupies drier lowlands, foothills, and agricultural edges.
Behavior
The great horned owl is a largely solitary, territorial, and predominantly nocturnal raptor whose behavior reflects both its adaptability and its position as an apex predator across most of the Americas. It is active mostly at night and during twilight, spending daylight hours roosting in dense cover or on sheltered perches. Its behavioral repertoire combines deliberate, efficient movement on the ground and in flight, strong territoriality, a spectrum of threat displays, and complex interactions with other species, including humans.
Locomotion and Flight
On the ground the great horned owl moves with a slow, deliberate gait, often described as slightly waddling, and it may walk or hop to reach prey or navigate nest ledges. Walking is generally limited and inefficient, but the owl readily climbs angled trunks or branches using its feet and wings, particularly in young birds that ascend trees before they can fly.

Once airborne, it shows powerful, deep wingbeats followed by short glides, holding its broad wings nearly horizontal and tucking the head, giving a compact, short-necked silhouette. Its wing structure allows low-speed maneuvering, slow quartering flights during hunting, and even brief hovering in strong winds. Silent flight is a key adaptation: modified feathers dampen sound, allowing close approach to prey.
Maintenance and Daily Activities
Self-maintenance includes frequent preening, especially after flight or in windy conditions, and systematic scratching of the head, facial disc, and nape using one or more forward claws. Individuals perform stretching sequences, extending wings and legs singly or together, and occasionally bathe by wading into shallow water and splashing vigorously.
Roosting sites vary widely, from conifers and deciduous canopies to cavities, cliffs, and human structures. Owls typically select sheltered, inconspicuous roosts that provide protection from weather and mobbing birds. Outside the breeding period they may roost wherever their nightly foraging ends, while males in the nesting season often use traditional daytime roosts near the nest. Sleep is light and easily interrupted; ear-tufts and posture shift frequently in response to surrounding sounds.
Social Structure and Interspecific Interactions
Great horned owls are strongly territorial and remain solitary for most of the year, pairing only during the breeding season. Mated pairs occupy the same territory but typically roost separately until courtship intensifies. After fledging, young siblings may travel together for several weeks before dispersing. Agonistic behavior includes a graded series of threat displays – from hissing, bill-clapping, and guttural calls to full wing-spreading, crouching, and, if pressed, striking with the feet. Intraspecific aggression can be severe and occasionally lethal, particularly during food shortages.
The species elicits intense mobbing from crows, ravens, jays, and various raptors. Corvids in particular pursue and harass daytime-roosting owls persistently, calling loudly and attracting additional birds. Red-tailed hawks often occupy similar landscapes and may nest surprisingly close to great horned owls, but smaller owls frequently abandon territories when this larger species is present. Although generally dominant, great horned owls may be harassed by falcons or hawks and rarely take advantage of these pursuits to capture a mobbing bird.
Predation on adults is extremely rare, but nestlings and fledglings can be taken by mammals such as coyotes, bobcats, raccoons, and red foxes, especially if they fall from nests prematurely or are weakened by poor food availability. Cannibalism and siblicide occur infrequently but are documented during periods of prey scarcity.
Interactions with Humans
Interactions with humans are shaped primarily by the owl’s strong territorial instincts during the breeding season. While the species generally avoids people, it may defend nests vigorously. Most attacks occur when hikers, researchers, or unsuspecting passersby approach too closely, often without noticing a well-hidden nest. Defensive strikes are rapid and silent, typically directed at the back of the head using powerful feet and long talons. Although injuries are uncommon, documented cases include deep scalp lacerations, torn ears, and individuals knocked off balance.
These aggressive responses are generally restricted to nesting adults, especially females, and do not reflect unprovoked aggression. Outside the breeding season, great horned owls tolerate human presence more readily and frequently occupy suburban parks, farmland edges, and semi-urban environments. Nevertheless, observers are advised to give potential nest sites wide berth, especially at dawn and dusk in early spring.
Breeding
The great horned owl is an early-season breeder across its range and follows a monogamous mating system. Pairs form long-term bonds, often lasting many years, and typically remain on the same territory year-round. Most individuals breed in their second year, and the species is normally single-brooded, although a replacement clutch may be laid if an early nest fails. Breeding timing varies widely with latitude and climate: egg-laying begins as early as late November in southern Florida and peaks from January to March across much of North America, with the latest clutches in northern regions laid into May.
Courtship
Courtship begins in autumn or early winter, marked by increased hooting, territorial advertisement, and duet calling. Males initiate most vocal activity, and pairs engage in synchronized hoots that reinforce pair bonds and territorial ownership.

Courtship displays include deep bowing, tail-bobbing, inflated white throat patches, and repeated approaches and retreats. Both sexes participate, often lowering or spreading their wings and engaging in mutual preening and bill-rubbing. Males frequently call from potential nest sites, drawing the female’s attention to suitable locations. Copulation typically occurs shortly after sunset and may follow prolonged sequences of calling and posturing.
Nesting
This species does not build its own nest, instead appropriating stick nests of hawks, crows, ravens, magpies, herons, or squirrels, as well as cavities, cliff ledges, rock niches, and an extraordinary variety of human-made structures. Nest placement is highly variable and depends almost entirely on the site constructed by the nest’s original builder. Common substrates include aspen and cottonwood trees in northern forests, juniper in arid regions, and cavities or ledges in desert canyons and quarries.
In urban and suburban settings, nests occur on artificial platforms, abandoned buildings, bridges, and even unconventional locations such as flowerpots or grain bins. Nests typically receive minimal modification – occasionally a sparse lining of bark strips, feathers, prey fur, or trampled pellets, but often nothing at all. Because reused stick nests deteriorate quickly, most are occupied for only a single season unless built on a sturdy foundation.
Egg Laying and Incubation
Clutch size ranges from one to four eggs, most commonly two. Eggs are nearly spherical, dull white, and coarse in texture. Females begin incubation immediately after the first egg is laid, resulting in strongly asynchronous hatching. Only the female incubates, maintaining near-continuous attendance; the male provisions her with prey throughout the night. Incubation lasts 30-37 days. Pairs can tolerate extreme winter conditions – eggs remain viable even during brief absences at temperatures well below freezing. Second broods do not occur except when pairs re-lay after an early nest failure.
Hatching and Parental Care
Owlets hatch altricial, sparsely covered in white down, and completely dependent on the female for brooding during their first two weeks. The male continues to supply prey, which the female tears into small pieces for the chicks. Young develop rapidly: eyes open at 9-11 days, mesoptile down replaces natal plumage within the first week, ear tufts appear around three weeks, and flight feathers break sheath by the second week. Nestlings regulate body temperature effectively by three to four weeks, reducing the need for brooding except in severe weather. They begin to climb to surrounding branches at five to six weeks and make their first short flights around 45-49 days.

Parental care remains intensive after fledging. Adults continue to feed young for several weeks, and fledglings often remain in the natal area throughout summer, gradually perfecting flight and hunting skills. Sibling interactions are usually peaceful but can include siblicide during severe prey shortages. By late autumn, juveniles disperse from the parental territory; most do not breed until at least their second year, although rare first-year breeding has been documented.
Lifespan
Great horned owls are among the longest-lived owls in the Americas, with most individuals surviving several years in the wild and a smaller proportion living well over a decade. Typical wild lifespans fall in the range of 8-13 years for territory-holding adults, though survival varies widely with food supply, habitat quality, and local pressures. Exceptional individuals have reached advanced ages: one wild owl survived to a minimum of 28 years 7 months, and another banded as a nestling lived 23 years 4 months. Under human care or long-term rehabilitation, longevity increases dramatically – a well-documented Cincinnati-area owl injured on a highway survived to 34 years 10 months in captivity.
Mortality and Survival
Nestling mortality is comparatively low for a large raptor, generally 5-7% in well-studied populations. Deaths in the nest most often result from predation by raccoons, opossums, crows, or ravens, along with starvation during prey shortages. If a young owl falls from a nest, it becomes vulnerable to foxes, coyotes, and other ground predators. In years of scarce prey, siblicide may occur, with older nestlings killing weaker siblings.
Survival after fledging is far more variable and highly sensitive to food availability. First-year survival in stable Midwestern populations averages around 68%, increasing to 75% in the second year and to over 85% in subsequent adult years. However, in northern ecosystems tied to the ten-year snowshoe hare cycle, annual survival can oscillate sharply: juvenile survival may drop from 80% in prey-rich years to under 25% after prolonged declines. Adult survival is also reduced during prey crashes, though territorial adults generally fare better than non-territorial “floaters,” whose annual survival may fall to 40% in the poorest years.
A broad range of natural and anthropogenic factors contribute to mortality. Juveniles and inexperienced birds are particularly susceptible to food scarcity, blood-sucking black flies, and parasitic infections such as Leucocytozoon. West Nile virus (WNV) has emerged as an important cause of illness and death in some regions, with infected owls showing neurological symptoms and high mortality rates, though population-level impacts remain uncertain. Other pathogens documented in this species include avian tuberculosis, aspergillosis, herpesvirus, and various protozoan and helminth infections.
Human-related causes form a major share of adult and juvenile mortality. Great horned owls are regularly admitted to rehabilitation centers after vehicle collisions, electrocutions, shootings, entanglement in barbed wire, and injuries from leghold traps. Additional cases involve damage from skunk spray leading to corneal opacity and impaired vision, or from dangerous prey – porcupine quills, for example, have caused fatal injuries. Despite these challenges, breeding adults in stable habitats generally show strong annual survival, underscoring the resilience of this widespread predator once they reach maturity.
Diet
The great horned owl is an exceptionally broad-niche predator and one of the most versatile feeders among North American raptors. It is primarily a nocturnal perch hunter, relying on acute night vision, sensitive hearing, and powerful, silent flight. Although most active from late evening through early morning, it readily adjusts its schedule, hunting at dawn, dusk, and even fully in daylight when energetic needs increase, especially during chick-rearing. Its diet spans an extraordinary range, from insects and small rodents to waterbirds, skunks, porcupines, and adult rabbits, reflecting a strongly opportunistic foraging strategy shaped by local prey availability.
Feeding and Foraging Behavior
Most hunting is performed from elevated perches overlooking open or semi-open habitats. The owl scans fields, meadows, wetland edges, agricultural landscapes, and woodland margins, using both vision and hearing to detect prey movements. It may also quarter low over grasslands or sagebrush, walk along the ground to stalk small mammals and invertebrates, or exploit human structures such as telephone poles, barns, and bridges as vantage points.
Effective strike distance can exceed 90 meters (295 feet) from a perch. Prey are captured with exceptionally strong feet, capable of delivering over 13 kilograms (29 pounds) of force, and are typically crushed or decapitated before swallowing. Small items are consumed whole, while larger prey are dismembered or carried back to the nest in pieces. When food is abundant, individuals may cache surplus kills, including snowshoe hares or large birds, and return over multiple days to consume them.
Major Prey Types
Across most of its range, mammals dominate the diet by biomass and number. Rabbits and hares, pocket gophers, voles, mice, rats, ground squirrels, muskrats, and various medium-sized mammals form the core prey base. Regionally, the species may also take skunks, raccoons, marmots, woodchucks, or even house cats. Birds constitute the second most important group and include waterfowl, coots, grebes, pigeons, corvids, galliforms, and other owls – sometimes including species similar in size or larger. Nestling birds of diurnal raptors and corvids are taken regularly at night.
Amphibians, reptiles, fish, and invertebrates contribute small but ecologically meaningful proportions, with some desert or arid-region populations incorporating high numbers of scorpions, beetles, or reptiles. Diet composition varies widely by region: for example, voles may dominate pellets in some interior habitats, while waterbirds form the bulk of prey on coastal islands.
Foraging Ecology and Selection
Prey selection reflects a combination of abundance, vulnerability, habitat structure, and energetic yield. Open microhabitats consistently increase detection and capture success, and prey taken often includes species that use exposed areas at night. In the boreal forest, snowshoe hares are strongly preferred when abundant; in arid regions, reptiles and large arthropods become more prominent. Large-bodied prey such as hares, rabbits, and waterfowl provide high energetic returns and may support rapid early-season breeding.

The owl generally selects larger individuals within a prey species when possible, but will shift to small mammals, rodents, or invertebrates when conditions require. The tendency to store prey at nests increases during the nestling stage or during winters with patchy food distribution.
Dietary Shifts and Prey Cycles
Great horned owls show marked functional responses to prey fluctuations. In the northern boreal forest, populations track the 10-year snowshoe hare cycle closely. At hare peaks, hares may constitute 75-98% of winter biomass; at troughs, they fall to near zero, replaced by ground squirrels, mice, voles, and various birds. These shifts have major demographic consequences: when hares decline, owls either switch to alternative prey, expand their foraging range, or undertake irruptive movements; failure to compensate often results in starvation or emigration. Similar patterns occur in the Great Basin, where jackrabbit cycles govern prey composition.
Seasonal dynamics also influence diet: in some regions, cottontails predominate in autumn, voles peak in spring, and birds increase during migration periods. Such flexibility underlies the species’ broad distribution and its ability to persist in diverse habitats, from boreal forest to desert shrublands.
Culture
The great horned owl holds a distinctive place in human culture, shaped by its imposing appearance, nocturnal habits, and widespread distribution. Across the Americas, this species has occupied both symbolic and practical roles in Indigenous cultures. Many Native nations associated the owl with strength, courage, or protective power. Others viewed its call as an omen linked to seasonal change, fertility, or the return of warm weather.
Among several tribes of the Great Plains and Southwest, owl feathers were incorporated into ritual garments or weaponry to invoke silence, stealth, or spiritual potency. In the Northeast, stories explained the owl’s nocturnal habits and dark plumage through creation myths. Some groups regarded the great horned owl as a conduit for wisdom or as a vessel for the spirits of the virtuous dead.
In parts of Central America, the species also plays a role in hunting traditions and folklore. Its call is sometimes interpreted as a sign that game animals are nearby, and in local tales the owl acts as a guide or messenger who aids hunters or reveals hidden quarry. Today the great horned owl remains a potent cultural emblem, reflected in its designation as the provincial bird of Alberta and in its enduring presence in art, storytelling, and conservation symbolism throughout its range.
Threats and Conservation
The great horned owl remains one of the most secure raptors in the Americas. Its global population is extremely large – estimated at roughly 5.7 million mature individuals, and long-term survey data show broad stability across most of its range. The species adapts readily to habitat change, occupies an enormous geographic area, and uses a wide range of nest sites. As a result, the IUCN Red List classifies it as Least Concern, with no indication of rapid decline. Local fluctuations do occur, especially in northern regions tied to prey cycles, but there is no evidence of a sustained global downturn.
Main Threats
Although resilient, great horned owls face several chronic human-related threats. Secondary poisoning from anticoagulant rodenticides is among the most significant: numerous studies report residues in the majority of tested individuals, and lethal exposures occur regularly even at low toxin concentrations. Occasional poisoning from organophosphates, organochlorines, and other environmental contaminants has also been documented.
Collisions and electrocutions represent another steady source of mortality. Birds are struck by vehicles, power lines, and other structures, or electrocuted when perching on distribution poles. Despite full legal protection, illegal shooting and trapping still occur in some regions, though these pressures are far lower than in the early twentieth century.
In northern latitudes, periodic prey crashes, especially the 10-year snowshoe hare cycle, produce sharp increases in juvenile mortality and trigger irruptive movements. Disease, including West Nile virus, avian tuberculosis, Aspergillus infections, and parasitism by blood-feeding flies and Leucocytozoon, further contributes to sporadic local losses.
Finally, as a powerful generalist predator, great horned owls can negatively affect other sensitive species. Predation on reintroduced peregrine falcons, spotted owls, terns, plovers, and other colonial or cavity-nesting birds is well documented.
Conservation and Management
Because populations remain robust, conservation actions focus more on management than on species recovery. Limiting the use of second-generation anticoagulant rodenticides, especially near natural areas, is widely recognized as a key mitigation measure. Reducing collision risks through better placement and design of power infrastructure also benefits local populations. In regions with few natural nest sites, artificial platforms or baskets successfully support breeding pairs. Targeted removal of individual owls is sometimes necessary where they consistently prey on endangered species, though such actions are used sparingly.
Otherwise, the species requires little direct intervention: its ability to thrive in fragmented, suburban, and agricultural landscapes allows it to persist where many other raptors decline.
Future Outlook
Given its ecological flexibility, large range, and steady global numbers, the great horned owl’s overall outlook is positive. Continued monitoring is important, especially in areas with high rodenticide use, increasing urbanization, or emerging diseases, but current data suggest that the species will remain widespread and abundant for the foreseeable future. Its future conservation needs will likely center on managing human-wildlife conflicts, reducing toxic exposures, and safeguarding the broader ecosystems that support its diverse prey base.
